ATI-1013 (Development)
Our lead molecule, a fully human, monoclonal antibody (mAb) with very high affinity and selectivity for nicotine has achieved preclinical proof of concept and entered development.
ATI-1013 was discovered by harnessing the power of the human immune system to generate high affinity antibodies to external antigens. ATI-1013 was isolated by screening over 50 million antibody producing blood cells (B-lymphocytes) isolated from donors who previously participated in experimental nicotine vaccine studies.
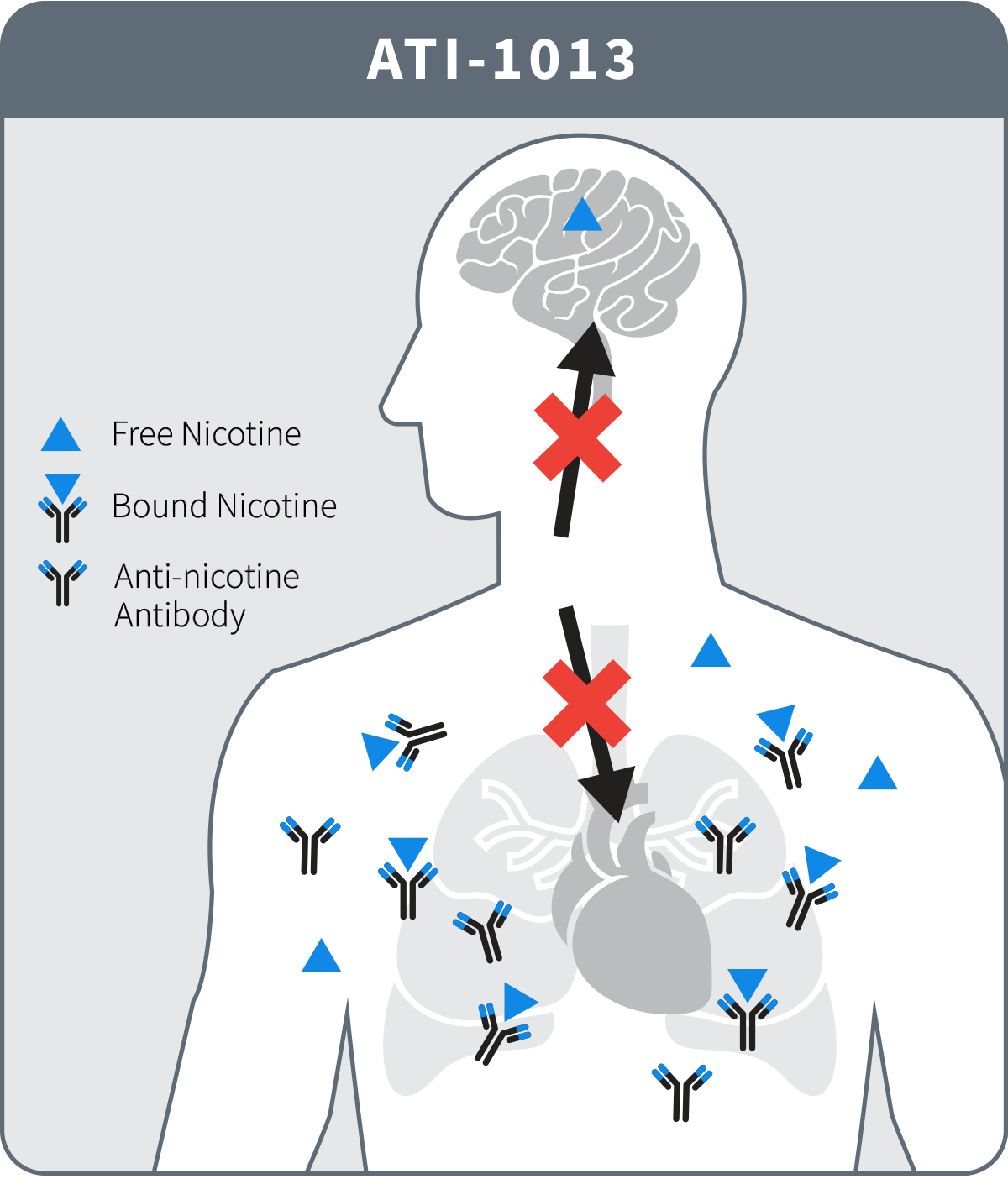
In contrast to small molecules like nicotine, antibodies are large proteins that cannot enter the brain but stay in the bloodstream. Their anti-nicotine action occurs because ATI-1013 binds nicotine and sequesters it in the blood, neutralizing its pharmacological actions at all of nicotine’s receptors found in the body. Additionally, since the antibody-bound nicotine is too large to cross the blood-brain barrier, ATI-1013 slows and reduces the amount of nicotine entering the brain thereby attenuating signaling of neuronal nicotinic acetylcholine receptors considered to be central to nicotine’s addictiveness. This innovative anti-nicotine approach circumvents side effects that accompany other pharmacotherapies for treating nicotine addiction. When given to animals prior to a nicotine dose, ATI-1013 reduces entry of nicotine from the blood into the brain by more than 90%. This antibody-induced sequestration of nicotine in the blood and not in the brain is expected to break the nicotine addiction cycle.
ATI-1013 also blocks nicotine from binding to its receptors outside of the brain, such as those in the adrenal gland and the vascular endothelium that are responsible for smoking-induced release of epinephrine and related substances that cause vasoconstriction, blood pressure elevation, and increased heart rate. Consequently, ATI-1013 possesses a unique dual anti-nicotine mechanism of action.

